Purple cabbage, like elderberry juice, is a natural pH indicator. That means that it changes colors depending when you add substances to it of varying pH levels. More acidic substances, like vinegar and lemon juice, turn it more pink. More basic substances, like dish detergent, turn it more blue.
We came up with this experiment in our own homeschool over ten years ago and it’s been a really fun activity that we’ve come back to many times over the years and introduced lots of other homeschooling friends too.
You have to do this with your kiddos. Not only is it educational, but it’s just insanely fun. 🙂
Here’s all you do.
1. Chop about 2 cups of purple cabbage and cover with boiling water. Put a lid on it and steep for about 10 minutes.

2. Strain out the liquid and reserve. It should be a deep blue-purple color, though it may be affected somewhat by your water.

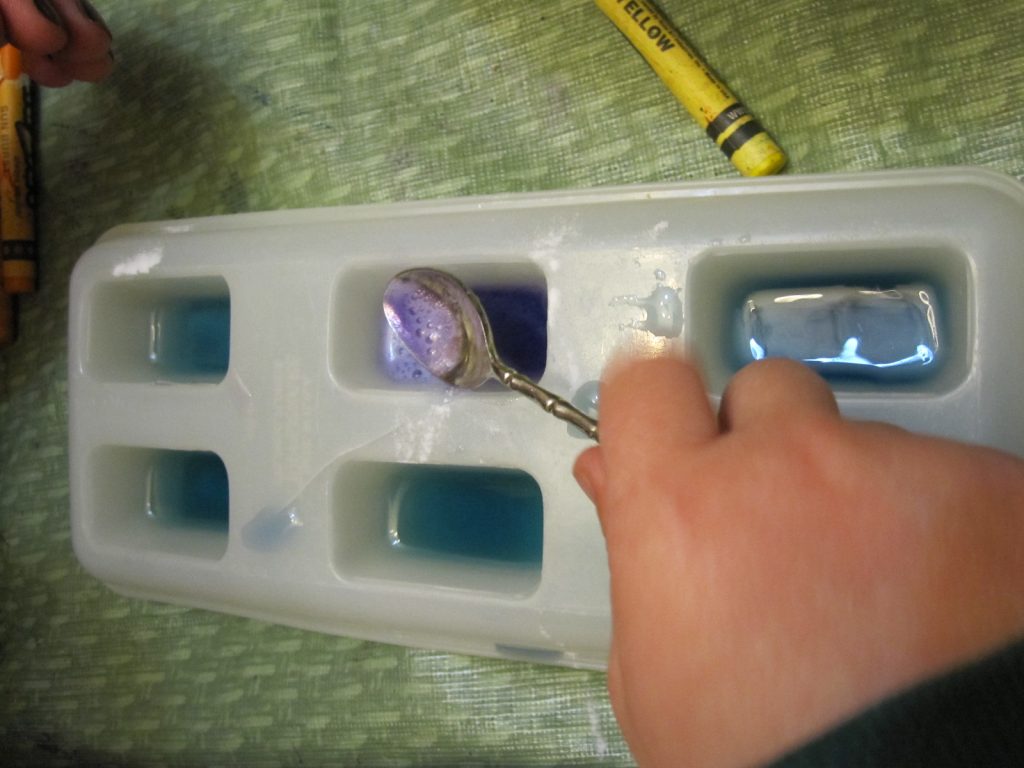
3. Put a little cabbage water in each compartment of a light colored ice cube tray or similar container with many sections. For little kids, a muffin tin works well.

4. Assemble a variety of substances (preferably white or clear) to test. For younger kids, put just a few in some larger dishes. Older kids can gather lots of test substances and use them directly from the containers.


5. Give each child a small spoon and an eye dropper and explain about the pH (potential of Hydrogen) scale and how pH indicators change color to reflect the pH value of substances (more pink for acidic, more towards green or almost yellow for base, though blue was generally as alkaline as we got). This site gives more background info (though they do a different experiment).

6. Let the kids add various substances and see what happens! Make sure you keep track of which substance was added to which compartment.

Check out this range of colors Victoria got from various substances!

(Substances used on top from left to right: hot sauce, white wine, lemon juice, witch hazel, alcohol, fingernail polish remover, hydrogen peroxide. On bottom: control spot with nothing added, baking powder, alum, salt, club soda, apple cider vinegar, baking soda)
Some of the best reactions were from the alum, hydrogen peroxide, hot sauce (2 drops and it was hot pink!), wine, club soda… okay, there were an awful lot of good substances, and even those that didn’t change much taught us something!
Next time I’d like to compare rainwater (or melted snow) to tap water to distilled water, plus see about detergent (which is supposed to be very alkaline).
But wait, there’s more! Once you’ve changed the colors of the liquid in each, challenge the kids to change it back! Ask them to think about what they can add to a hot pink to make it blue again, for instance. In a few cases, you end up with a reaction (notably baking soda and vinegar ending up in one compartment).
Obviously, don’t put out substances that can react violently together (like bleach and ammonia) but I wouldn’t recommend putting those out to begin with. The harshest solution we dealt with was probably the fingernail polish remover, which only Victoria and Anna used.
When younger children are doing this activity, I only put out very benign materials like baking soda, lemon juice, baking powder and vinegar. Even with just these they can change the colors back and forth and make reactions. We’ve had some of our younger kids stay busy with a muffin tin, cabbage water, bowls of baking soda and vinegar and an eye dropper all afternoon.
Discuss pH while you do this activity — how shampoo is supposed to be pH balanced so it doesn’t sting eyes, how some hydrangeas are pink in acidic soil and blue in alkaline, and so on.
~~~~~~~~~
Update: 3/3/17 This project is now featured (along with 51 others) in my new nature studies book: A Magical Homeschool: Nature Studies (52 Wonderful Ways to Use Nature Studies in Every Season to Teach Science, Math, Art and More)
my new nature studies book: A Magical Homeschool: Nature Studies (52 Wonderful Ways to Use Nature Studies in Every Season to Teach Science, Math, Art and More)
It’s available in Kindle version or print version (which also includes nature journal pages, logs for bird sightings, garden/foraging records and more). 🙂 (affiliate link)






One thought on “The Purple Cabbage pH Experiment”-
10 Wonderful Ways to Learn with Nature in March – Alicia Bayer
(March 7, 2018 - 8:09 pm)[…] is one of those science projects that’s so much fun the kids will ask to do it again and again. Use purple cabbage juice to test pH of household liquids and make all sorts of […]